INL, as one of partners in the Department of Energy (DOE)-sponsored Battery500 consortium, is developing the next-generation battery technologies with higher energy, lighter weight, and long cycle life to power future electric vehicles. The overarching goal of the Battery500 consortium is to build a battery with a specific energy of 500 watt-hours per kilogram, which is more than twice the specific energy of today's typical electric-vehicle (EV) battery. Li metal-based batteries, such as Li-sulfur and high-nickel-content nickel-manganese-cobalt (NMC) batteries hold great potential due to their high specific energy. However, they suffer from short cycle life. INL addresses these issues by building stable solid electrolyte interlayer (SEI) on anode surfaces, rationally designing cells, optimizing use conditions, and increasing utilization of cathode materials as well as improving electrode kinetics. INL has established strong expertise and capability in discovering and developing novel technologies, and electrode architecture design and interfacial engineering for Li-S and Li-NMC batteries. These technologies—and associated electrode active-material and architecture characterizations using physicochemical and electrochemical techniques—are aiding the Battery500 Consortium to achieve project goals. Our latest publications on this topic can be found below.
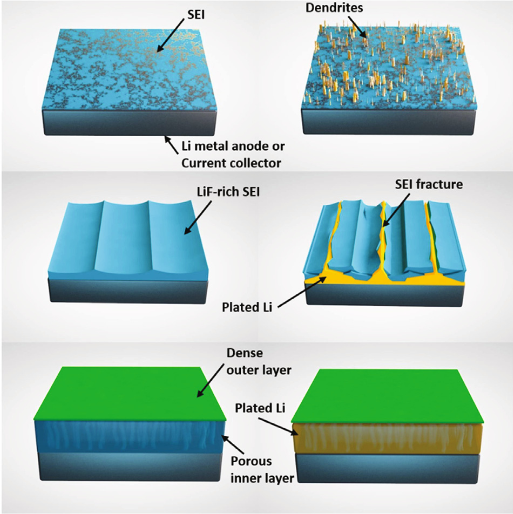
INL demonstrated a bi-layer SEI structure, constructed of an interconnected, tortuous, porous LiF-rich layer in contact with lithium and a dense in-situ formed inorganic-rich layer on top of the porous structure, showing enhanced anode stability. The resulting artificial SEI was a LiF-rich, porous structure, where an increased number of Li/LiF interfaces for lithium nucleation are made available, thus reducing local volume fluctuations, improving the flexibility of the SEI, as well as decreasing anode resistance because of faster Li+ diffusion along such interface. The interconnected and tortuous pores improve the Li+ flux distribution and mechanically suppresses dendrite growth on the Li-metal surface. The Li- metal with the bi-layer SEI showed extended cyclability in symmetric cells and pouch cells using both sulfur and LFP cathodes. This rational design of SEI opens the new opportunity to improve the stability of the Li-metal anode and unlocks a plausible route for high-energy metal-based batteries, such as Li, Na and K metals. Schematic showing the initial structure and failure mechanism for (top) Li-metal and (middle) dense LiF artificial SEI, as well as (bottom) the plating mechanism fora bi-layer dense/porous artificial SEI.

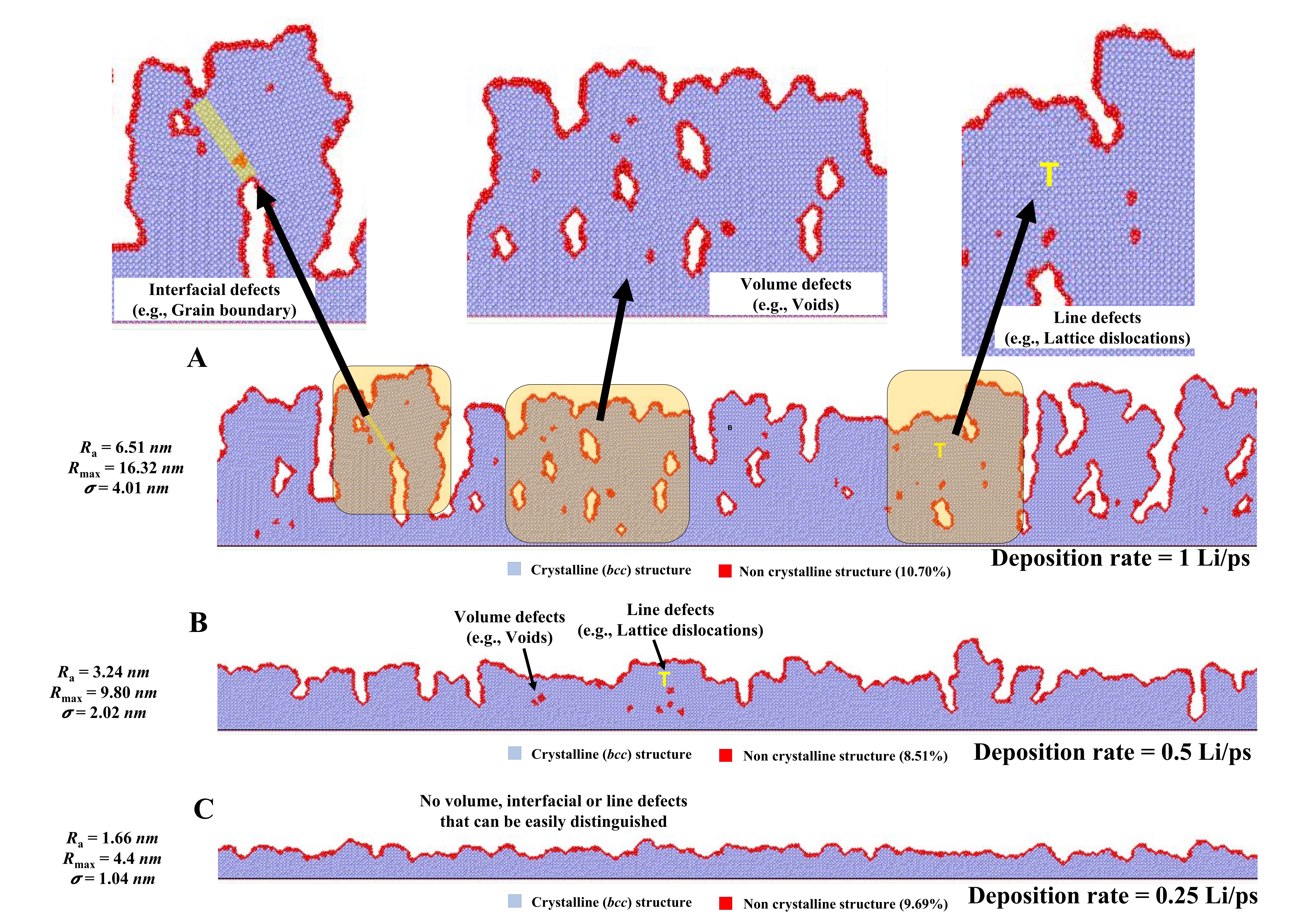
![]()
![]()
INL and the University of California at San Diego team correlate the crystallinity of Li nuclei with subsequent growth of the nanostructure and morphology and provide strategies to control and shape the mesostructure of Li metal to achieve performance in rechargeable Li batteries. For more, please visit the article Glassy Li metal anode for high-performance rechargeable Li batteries.
Understanding lithium−sulfur battery performance with lean electrolytes is highly desirable. INL researchers developed methods to synthesize a nanoporous carbon host, decorated with Ni3S2@Ni particles. Such a cathode delivers enhanced specific capacities with extended cycling life in lean electrolytes due to the dual functions of the Ni3S2 shell, which can both facilitate reaction kinetics and promote electrolyte wetting. This work highlights a strategy to rationally design cathodes for high-energy lithium−sulfur batteries.
Publications
2024
2023
2022
2021
2020
2019
2018
Research Contact: Tanvir Tanim - Phone: (208) 526-5713 - Tanvir.Tanim@inl.gov
Eric Dufek - Phone: (208) 526-2132 - Eric.Dufek@inl.gov